Category: Medications - Page 2
How to Address Allergies to Inactive Ingredients in Generic Medications
Learn how to spot allergens in generic drugs like lactose, gluten, and dyes, and why the inactive ingredient list matters for your safety.
Read moreTapering Opioids Safely: How to Reduce Side Effects and Withdrawal Symptoms
Learn how to safely reduce opioid doses to avoid withdrawal symptoms and side effects. Evidence-based strategies from the CDC and clinical studies show that slow, patient-led tapering improves pain, function, and safety.
Read moreHow Paragraph IV Patent Challenges Speed Up Generic Drug Entry
Paragraph IV under the Hatch-Waxman Act lets generic drug makers challenge brand patents to speed up affordable drug access. It’s how most generic drugs enter the market-and why prices drop so sharply.
Read moreHow to Read Medication Guides for Risk and Monitoring Advice
Learn how to read Medication Guides to spot life-saving risk warnings and monitoring instructions for your prescriptions. Know exactly what to watch for, when to act, and what to do if you don’t get the guide.
Read moreHow to Prepare for Pediatric Procedures with Pre-Op Medications
Learn how to safely prepare your child for surgery using pre-op medications, fasting rules, and anxiety-reducing strategies backed by pediatric anesthesia guidelines. Reduce fear, prevent complications, and ensure a smoother experience.
Read moreFive Essential Rules for Taking Medication Safely
Learn the five essential rules for taking medication safely to avoid dangerous errors. From knowing your pills to checking doses and timing, these simple steps can prevent hospital visits and save lives.
Read moreShared Decision-Making Scripts for Side Effect Trade-Offs in Medication Choices
Shared decision-making scripts help patients and doctors weigh medication side effects together, leading to better adherence, fewer regrets, and more personalized care. Learn how clear communication and real numbers make a difference.
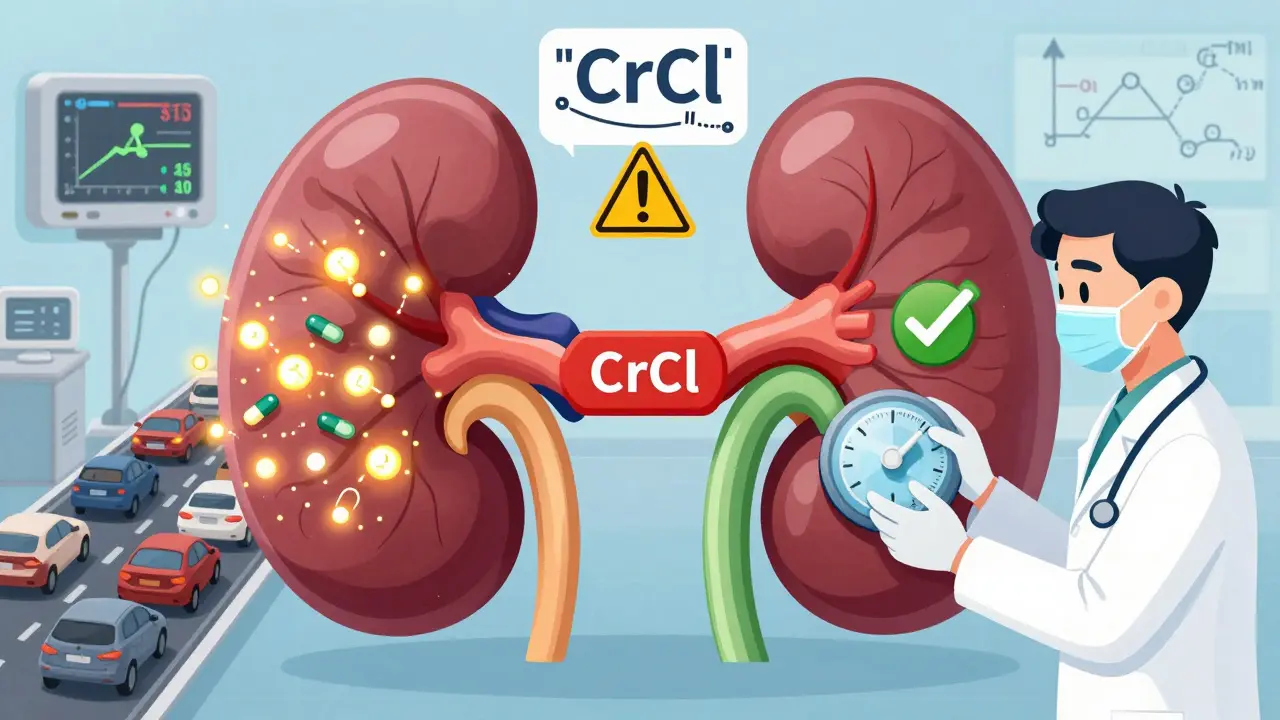
Read moreRenal Dosing of Antibiotics: Avoiding Toxicity in Kidney Disease
Renal dosing of antibiotics is critical to avoid toxicity in kidney disease. Learn how creatinine clearance guides dosing, why guidelines differ, and how to prevent life-threatening errors in clinical practice.
Read moreHow Family History and Genetics Affect Your Response to Generic Drugs
Family history and genetic factors play a major role in how your body responds to generic drugs. Understanding your genes can prevent dangerous side effects and help you get the right dose - not just the right drug.
Read moreSGLT2 Inhibitors and Diabetic Ketoacidosis Risk: Symptoms, Causes, and Prevention
Learn how SGLT2 inhibitors for type 2 diabetes can cause diabetic ketoacidosis even with normal blood sugar. Understand symptoms, risk factors, and how to prevent this serious complication. Includes latest guidelines and real-world data on managing this rare but dangerous side effect.
Read moreDe facto combinations: Why taking separate generics instead of FDCs is common - and risky
Many patients take separate generic drugs instead of fixed-dose combinations (FDCs) for cost or dosing reasons. But this practice, called a de facto combination, carries hidden risks like lower adherence and untested drug interactions. Learn when it's safe - and when it's not.
Read moreLevodopa and Antipsychotics: How Opposing Dopamine Effects Worsen Symptoms
Levodopa and antipsychotics have opposing effects on dopamine, making it dangerous to use them together. This article explains how this interaction worsens symptoms in Parkinson’s and schizophrenia patients, and what new treatments are emerging.
Read more