Tag: narrow therapeutic index
Prior Authorization for NTI Drugs: Why Insurers Require Brands and How to Navigate It
Navigating prior authorization for NTI drugs can be confusing. Learn why insurers require brand-name versions, which drugs are affected, and strategies to get coverage quickly in 2026.
Read moreNTI-specific substitution laws: which states have special rules
Twenty-seven states have special rules blocking or restricting generic substitution for Narrow Therapeutic Index (NTI) drugs like warfarin and levothyroxine. These state laws override FDA guidelines and create a patchwork of requirements - from mandatory consent to outright bans.
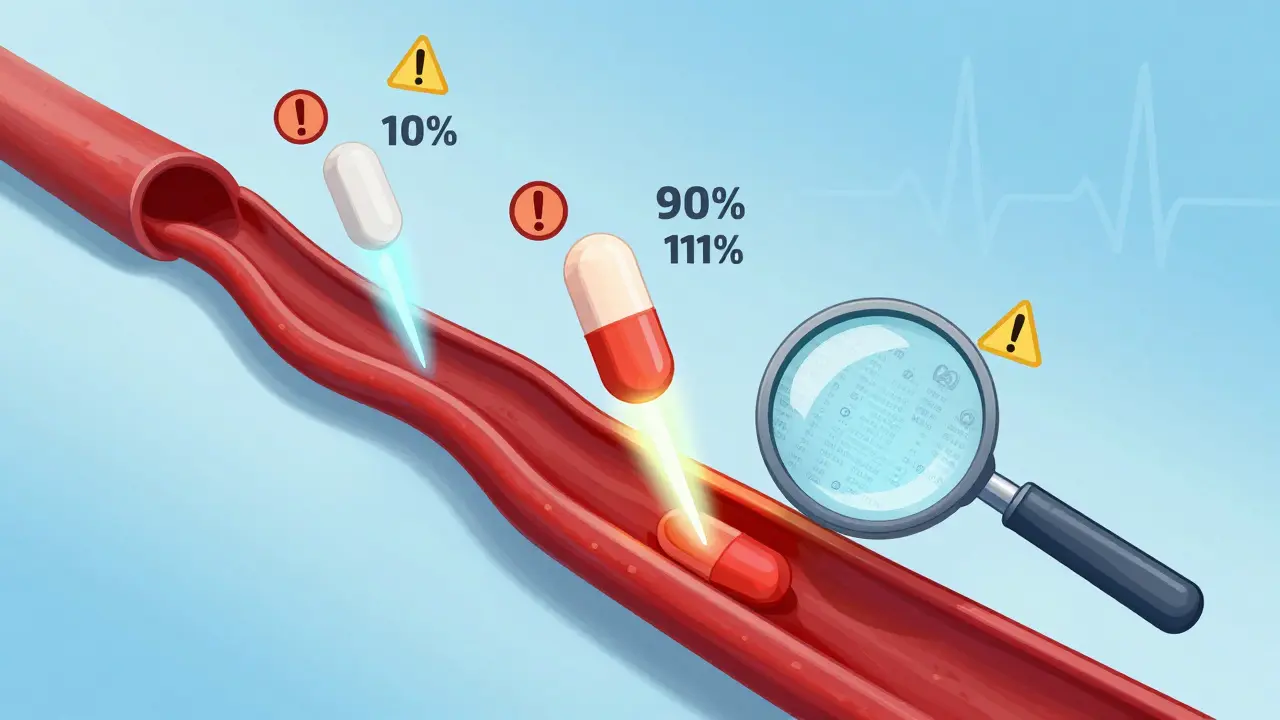
Read moreFDA Bioequivalence Standards for NTI Drugs: Special Requirements Explained
The FDA applies strict bioequivalence rules to narrow therapeutic index (NTI) drugs like warfarin and phenytoin, requiring tighter limits (90-111%) and lower variability than standard generics to ensure safety and efficacy.
Read more